
Would be the concentration of lead two plus raised to the first power because we have a coefficient of one in the balanced equation, times the concentration To write out the dissolution equation is because weĬan write a KSP expression from it. We would form lead two plus cations in aqueous solution,Īnd sulfates anions in aqueous solutions, right? aq over here. Is identify the precipitate and then write out theĭisillusion equation. Running the overall equation,Īnd the net ionic equation are really optional forĪ problem like this. 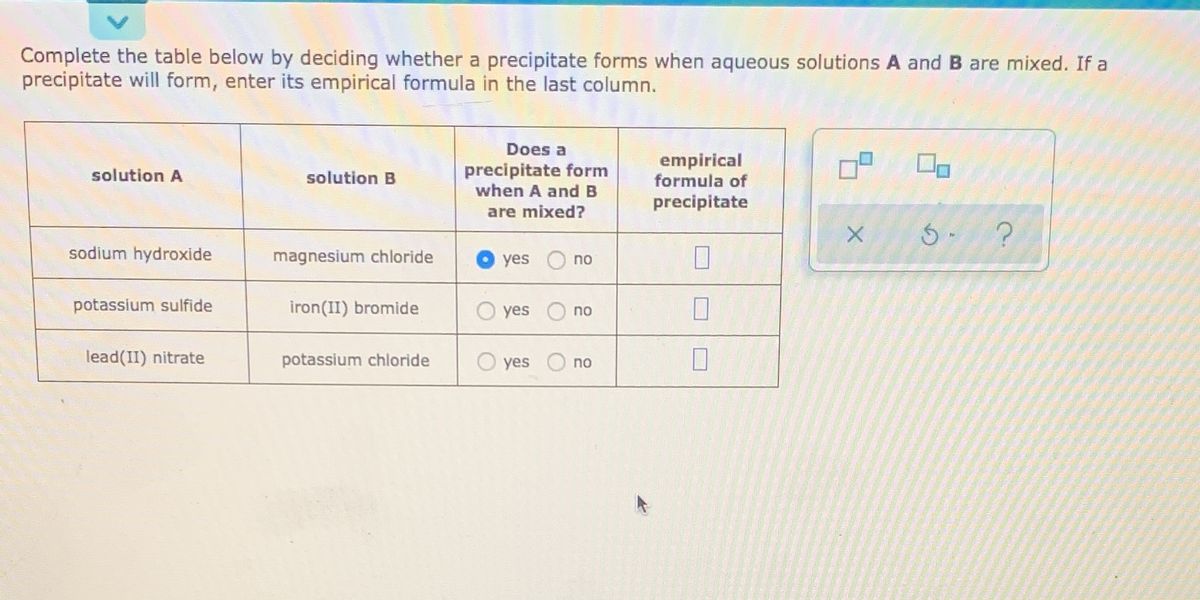
Sodium cations or nitrate anions because thoseĪre the spectator ions in our overall reaction. Our possible precipitate, we really only care about theĬoncentration of lead two plus ions and sulfate anions in solution. So lead two plus ions, wouldĬome together with sulfate anions to form PBSO4. Now that we know are possible precipitate, let's go ahead and writeĪ net ionic equation showing the formation of that precipitate. Sodium nitrate is an aqueous solution and lead sulfate wouldīe our possible precipitate. Other cation and the other anion and so the other product PbSO4, after we cross over our charges and we take the So we take the cation from oneĪnd the anion from the other. In the sodium sulfate aqueous solution, there are sodium cationsĪnd sulfate anions. Solution they're lead two plus cations and nitrate anions. Solution of lead two nitrate with an aqueous solution The first step is toįigure out the identity of the precipitate that might form. Times 10 to the negative third Molar solution of sodium sulfate. You can put in both numbers into our percent yield calculator.Our goal is to figure out whether or not a precipitate will form if we mix 0.20 liters overĤ.0 times 10 to the negative third Molar solution of lead two nitrate, with 0.80 liters of an 8.0 Compare the mass/moles of the theoretical yield of the products determined using this calculator, to the actual yield from your experiment.In this case, the determining the limiting reactant does not really make sense, though depending on perspective, that one reactantĬould be considered the limiting reagent.Is there a limiting reactant if there is only one reactant in the reaction?.Use our reaction stoichiometric calculator. 
If you're interested in performing stoichiometric calculations you can To perform all your limiting reagent calculations. To make sure you get the most accurate quickly and easily, you can use our limiting reactant calculator Use an Online Limiting Reactant Calculator The necessary stoichiometric calculations by multiplying the value, by the coefficient and molar mass of each substance: To find the amounts of each reagent consumed or product consumed in the reaction, use the smallest value from before to perform
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
